Site sections
Editor's Choice:
- Why at home may there be a need for measuring water resistance?
- Alteration of "Khrushchev" dvushki almost treshku
- Paint the wallpaper with your own hands
- Installation options for drywall in the bathroom
- Court decision on recovery from the management company of the amount of damage to the gulf of the apartment
- Living room and children in the same room: options for partitions
- Top sofa upholstery rating: customer reviews
- Expansion joints in buildings
- Chaber - what is it and its purpose
- Sharpening wood cutters: manual work, using grinding wheels and a grinding machine
Advertising
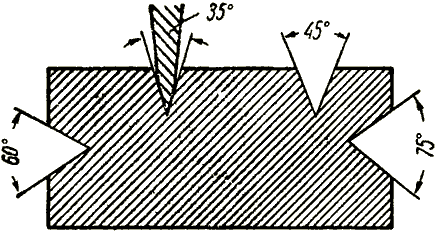
Characteristics of the cylinders used in the gas-flame processing of metals.For storage and transportation of compressed, liquefied and dissolved gases under pressure above atmospheric, steel cylinders of various capacities are used: from 0.4 to 55 liters. According to GOST 949-73 *, cylinders are made from seamless carbon or alloyed steel pipes with nominal pressure up to 200 kgf / cm 2. For some liquefied gases (propane, butane, their mixtures, etc.), and sometimes dissolved acetylene at a working pressure not higher than 30 kgf / cm 2, welded cylinders are used. Oxygen cylinders (fig. 28). Gaseous oxygen is stored and transported in steel cylinders under a pressure of 150 kgf / cm 2. Fig. 28 Oxygen balloon: 1-support shoe, 2-body, 3 - ring, 4 - shut-off valve, 5 - safety cap For approximate determine the amount of oxygen in the balloon, you can use the formula V to = V b P to, where V to - the amount of oxygen in the cylinder, l; V b - water tank capacity, l; P to the pressure of oxygen in the cylinder gauge, kgf / cm 2. Thus, in a full oxygen cylinder, the amount of oxygen is: 40X150 = 6000 l, or 6 m 3 (at atmospheric pressure). Acetylene Cylinders(fig. 29). Acetylene, in contrast to compressed gases, is stored and transported in a dissolved state. Acetylene cylinders are manufactured according to GOST 5948-60 and have the same dimensions as the oxygen ones. In addition to seamless cylinders from seamless pipes, welded cylinders of type BAS-1-58, made of carbon or low-alloy steel, are also used. Fig. 29. Acetylene Cylinders: a - seamless, b - welded BAS-1-58; 1 - body, 2 - stop valve, 3 - safety cap, 4 - gas cushion, 5 - porous mass with acetone, 6 - support shoe Inside the acetylene cylinder is a porous mass with acetone 5. The porous mass is a granular activated charcoal with a grain size of 1-3.5 mm brand BAU (GOST 6217-74). 290-320 g of activated charcoal is introduced into 1 liter container. Acetone (CH 3 COCH 3) is introduced into the balloon in the amount of 225-300 g per 1 liter of the container capacity. It permeates the porous mass and, when the cylinders are filled with acetylene, dissolves it well. The amount of acetylene in the cylinder in the filler plants is determined by weighing it before and after filling. For an approximate determination of the amount of acetylene in the balloon, you can use the formula V a = 7V b P a, where V a - the amount of acetylene in the cylinder, l; 7 - coefficient taking into account the amount of acetone and the solubility of acetylene; V b - water tank capacity, l; P and - the pressure of acetylene in the cylinder gauge, kgf / cm 2. So, in a full acetylene cylinder, the amount of acetylene gas is: 7X40X19 = 5320 or 5.32 m 3 (under normal conditions). Cylinders for liquefied gases(Fig. 30). For propane and propane-butane mixtures, welded cylinders are used. Cylinders with a capacity of 50 liters (for 23 kg of gas), an outer diameter of 309 mm, a wall thickness of 4.5 mm and a height of 950 mm are most widely used. The mass of such a cylinder is 35 kg, the working pressure in it is 16 kgf / cm 2.
Fig. thirty. Propane tank: 1 - body, 2 - bottom, 3 - support shoe, 4 - washers, 5 - upper sphere, 6 - valve, 7 - cap, 8 - cylinder passport plate The valve is the closing device for cylinders when filling, storing and consuming gases from them. Some data on the cylinders are given in Table. 13. 13. Some data on cylinders used in the flame treatment of metals
Acetylene balloon device Acetylene balloon is a universal container for the storage and transportation of acetylene. The cylinder body is made of seamless pipes according to GOST 949-73. A shoe is mounted on the lower part of the body in a hot condition, which gives stability to the cylinder in an upright position. In the upper spherical part of the neck valve is screwed, designed for filling and selection of gas. In the off position, the valve is a locking device. The cylinders are equipped with VBA-1 valves according to TU 26-05-527-82 (with a membrane sealer) or BA-I according to TU 6-21-23-84 (with ebonite sealer). A threaded ring is pressed on the outer part of the neck to tighten the safety cap. At the place of transition of the cylindrical part of the balloon to the spherical, the following data are stamped:
The cylinders must be painted white with the exception of the place of branding, which must be covered with a colorless varnish and circled in a red frame. On the cylindrical part of the container should be the inscription "ACETYLENE", applied in red paint. The color of the cylinders and the inscription on them can be made of oil, enamel or nitrokraska. The inscription on the cylinders must be at least 1/2 of a circle, and the letters must be at least 60 mm high. Acetylene cylinder filled with porous filler and filled with acetone The role of the porous filler:
Depending on the porous filler, acetylene cylinders are divided into cylinders with a bulk porous mass (BAU-A coal) and cylinders with a cast porous mass (LPM). Coal BAU-A is a black grain without mechanical impurities, produced according to GOST 6217-74. The cast porous mass is a gray cast porous block, produced according to TU 6-21-38-85 "Cylinders for dissolved acetylene with a cast porous mass". The weight of the stuffed porous mass is 280-310 g per 1 liter of the capacity of the cylinder body or 30% of its volume. The molded porous mass of LPM TU 6-21-38-85 is formed as a result of a hydrothermal reaction between silicon dioxide, calcium hydroxide and additives at elevated pressure and temperature directly in the cylinder, as a result of which a continuous molded porous block is formed. It is impossible to allow pressure in acetylene cylinders, which significantly exceeds 25 kg / cm2, due to safety conditions. As a result, the porous mass is impregnated with acetone, which significantly increases gas selectivity, as it is a good solvent for acetylene. For acetonation of cylinders used technical acetone GOST 2768-84 grade 1. Acetone is a colorless, flammable liquid with a characteristic odor. Auto-ignition temperature 465? С. The mixture of vapors of acetone with air is explosive: the explosive limits in volume fractions of acetone are the lower limit of 2.2; top - 13. Liquid acetone causes skin irritation. Acetone fumes cause irritation and upper respiratory disease. Acetone is introduced into the balloon at the rate of 225-230 g per liter of the container capacity. The volume occupied by acetone in the balloon is 25-30%. To avoid an excessive increase in pressure in the cylinder, a portion of the porous mass should not be filled with acetone. In the remaining empty volume (gas cushion) is compressed acetylene gas, saturated with acetone vapor, this volume is 16%. Allowable pressure of acetylene in cylinders depending on temperature: Features of operation of cylinders with BAU-A and LPM The principal difference in the operation of acetylene cylinders with BAU-A and LPM coal is that, when discharging and filling cylinders, it is not recommended to put at the same time on the discharge or filling ramp cylinders with BAU-A coal per 5 kg of acetylene and LPM at 7 kg of acetylene in 40 liter bottle. With simultaneous filling of cylinders designed for 5 and 7 kg of acetylene in a cylinder, it is almost impossible to achieve a gas withdrawability of 7 kg of acetylene in a cylinder with LPM, since cylinders filled to 5 kg increase the pressure in the ramp and prevent further filling of cylinders by 7 kg of acetylene. With the simultaneous discharge of cylinders with different gassability, acetylene can flow from cylinders with greater gaseous collection to cylinders, with a lower content of acetylene. Cylinder requirements The main reasons for which cylinders are not allowed to be filled:
In cylinders suitable for filling, it is necessary to measure the residual gas pressure, it should be no more than 1 kgf / cm 2. After measuring the residual pressure, the balloon is weighed on the balance to determine the amount of missing acetone. External examination is necessary to establish the state of the gaskets in the annular recesses of the valves, to replace worn out and only after that the cylinder can be attached to the filling ramp. New acetylene cylinders and cylinders, received from the consumer with a residual pressure below 0.5 kgf / cm 2, must be rinsed with acetylene by triple filling to a pressure of 20 kgf / cm 2 and subsequent discharge to a residual pressure of 0.5 kgf / cm 2. All acetylene cylinders are subject to periodic inspection every 5 years (the next examination expires on the 15th day of the month in which the test was conducted). The test is carried out with a nitrogen pressure of 35 kgf / cm 2. The condition of the porous mass is checked after 24 months and with each survey. Introduction Acetylene (C 2 H 2) is a chemical gaseous compound of carbon with hydrogen, without color, with a faint ethereal odor and sweetish taste. Acetylene in gas welding production is most prevalent due to the important qualities for welding (high flame temperature, high heat of combustion). So, with the decomposition of 1 kg of acetylene, 8473.6 kJ of heat is released. This is the only gas whose combustion is possible in the absence of oxygen (or oxidizer in general). Heat release during acetylene combustion is caused by the following processes:
Acetylene is lighter than air, the mass of 1 m 3 of acetylene at a temperature of 20 ° C (273 K) and normal atmospheric pressure is 1.09 kg. At normal pressure and temperature from –82.4 ° С (190.6 K) to –84.0 ° С (189 K), acetylene becomes liquid, and at –85 ° С (188 K) it hardens, forming crystals . Technical acetylene is available in two types: dissolved and gaseous. Dissolved technical grade A acetylene is intended for powering lighting installations, technical grade B acetylene dissolved and technical acetylene gas are intended as a combustible gas in the flame treatment of metals. Technical acetylene is obtained from calcium carbide by decomposition of the latter with water. At the same time, harmful impurities that contaminate acetylene pass from calcium carbide to acetylene: hydrogen sulfide, ammonia, phosphoric hydrogen, siliceous hydrogen. These impurities can degrade the properties of the deposited metal and are therefore removed from the acetylene by washing in water and chemical cleaning. Particularly undesirable impurity of phosphorous hydrogen, the content of more than 0.7% in acetylene increases the explosiveness of the latter. Acetylene propertiesThe main properties of acetylene are shown in table 1.
In terms of physico-chemical parameters, technical acetylene should comply with the standards specified in Table 2.
Acetylene solubilityAcetylene gas can dissolve in many liquids. Data on the solubility of acetylene in some liquids at atmospheric pressure and a temperature of 15 ° C are given in table 3. The solubility of acetylene in liquids increases with decreasing temperature. Data on the solubility of acetylene in acetone at different temperatures are given in table 4. Dissolved acetylene is called acetylene, which is in a balloon filled with a porous mass impregnated with a solvent — acetone. Artificial cooling of cylinders accelerates the process of filling them. In the pores of the porous mass, acetylene is dissolved in acetone. When opening the cylinder valve, acetylene is released from acetone as a gas. Dissolved acetylene is intended for its storage and transportation. Acetylene explosion hazardWhen using acetylene it is necessary to consider its explosive properties. It is the only gas widely used in industry, whose combustion and explosion are possible even in the absence of oxygen or other oxidizing agents. The autoignition temperature of acetylene depends on the pressure (Table 5). Increasing the pressure significantly reduces the auto-ignition temperature of acetylene. Particles of other substances present in acetylene increase the surface of its contact and thereby reduce the autoignition temperature at atmospheric pressure to the following values, ° C (K):
If acetylene is slowly heated to a temperature of 700–800 ° C (973–1073 K) at atmospheric pressure, it is polymerized, at which molecules condense and form more complex compounds: benzene C 6 H 6, styrene C 8 H 8, naphthalene C 10 H 8, toluene C 7 H 8, etc. Polymerization is always accompanied by the release of heat and with the rapid heating of acetylene can turn into its self-ignition or explosive decomposition. If, when compressing acetylene in a compressor to a pressure of 29 kgf / m 3 (2.9 MPa), the temperature at the end of this process does not exceed 275 ° C (548 K), then ignition does not occur, which allows filling cylinders with acetone for long-term storage and transportation. With increasing pressure, the temperature at which the polymerization process begins, decreases (Fig. 1). In practical use of acetylene, it is permissible to heat it to the following values of temperature, ° С (K):
One of the important indicators of the explosiveness of combustible gases and vapors is the ignition energy. The smaller this value, the more explosive this substance. The values of the ignition energy of acetylene (under normal conditions): with air - 19 kJ; in oxygen - 0.3 kJ. Water vapor serves as a phlegmatizer for acetylene, i.e. its presence significantly reduces the ability of acetylene to ignite in the presence of random sources of heat and explosive decomposition. According to current regulations for acetylene generators, in which acetylene is always saturated with water vapor, the limiting overpressure is 150 kPa, and the absolute - 250 kPa. At atmospheric pressure, a mixture of acetylene with air is explosive, if it contains 2.2% acetylene or more, a mixture with oxygen — 2.8% acetylene or more (the upper limits of the concentration of acetylene for its mixtures with air and oxygen do not exist, since sufficient ignition energy can explode and pure acetylene). Getting acetyleneIn industry, acetylene is produced by the decomposition of liquid combustible, such as oil, kerosene, by exposure to an electric arc discharge. A method of producing acetylene from natural gas (methane) is also used. A mixture of methane and oxygen is burned in special reactors at a temperature of 1300–1500 ° C. From the resulting mixture, concentrated acetylene is extracted with a solvent. Industrial production of acetylene is 30–40% cheaper than that of potassium carbide. Industrial acetylene is pumped into cylinders, where there is in the pores a special mass dissolved in acetone. In this form, consumers receive balloon industrial acetylene. The properties of acetylene do not depend on the method of its production. The residual pressure in an acetylene cylinder at a temperature of 20 ° С should be 0.05–0.1 MPa (0.5–1.0 kgf / cm 2). The working pressure in a filled cylinder must not exceed 1.9 MPa (19 kgf / cm 2) at 20 ° C. For the preservation of the filling mass, acetylene cannot be removed from a cylinder at a speed of 1700 dm 3 / h. Let us consider in more detail the method of producing acetylene in a generator of calcium carbide. Calcium carbide is produced by fusing coke and quicklime in electric arc furnaces at a temperature of 1900–2300 ° С, at which the reaction proceeds: Ca + 3C = CaC 2 + CO Molten calcium carbide is drained from the furnace into a mold where it cools. Then it is crushed and sorted into pieces ranging in size from 2 to 80 mm. The finished calcium carbide is packaged in hermetically sealed calcium should not be more than 3% of particles less than 2 mm in size (dust). According to GOST 1460-81 sizes (granulation) of calcium carbide pieces are established: 2 × 8; 8 × 15; 15 × 25; 25 × 80 mm. When interacting with water, calcium carbide releases acetylene gas and forms slaked lime in the residue, which is a waste. The reaction of calcium carbide decomposition with water occurs according to the following scheme: From 1 kg of chemically pure calcium carbide it is theoretically possible to obtain 372 dm 3 (liter) of acetylene. Practically due to the presence of impurities in calcium carbide, the output of acetylene is up to 280 dm 3 (liters). On average, to obtain 1000 dm 3 (liters) of acetylene consumes 4.3–4.5 kg of calcium carbide. Carbide dust when wetted with water decomposes almost instantly. Carbide dust can not be used in conventional acetylene generators, designed to work on lumpy calcium carbide. For the decomposition of carbide dust, special-purpose generators are used. For cooling acetylene during the decomposition of calcium carbide. Also used from 5 to 20 dm 3 (liters) of water per 1 kg of calcium carbide. Apply also the "dry" method of decomposition of calcium carbide. For 1 kg of finely crushed calcium carbide, 0.2–1 dm 3 (liter) of water is fed into the generator. In this slaking process, lime is not obtained in the form of liquid lime mud, but in the form of dry scum, the removal, transport and disposal of which is greatly simplified. Transportation and storageTechnical acetylene gas is transported through pipelines from seamless steel pipes in accordance with GOST 8731 and GOST 8734. The pressure of acetylene in the pipeline should be no more than 0.15 MPa (1.5 kgf / cm 2). Coloring of pipelines - in accordance with GOST 14202. The gas pressure in the pipeline should be measured with a pressure gauge of accuracy class 2.5 according to GOST 8625, on the dial of which the inscription “Acetylene” should appear. Technical dissolved acetylene is filled with steel cylinders for dissolved acetylene with a porous mass (active carbon or cast porous mass) and acetylene. Cylinders must be equipped with special types of valves intended for acetylene cylinders. The gas pressure in the cylinder should be measured with a manometer of accuracy class not lower than 4 according to GOST 8625. The gas temperature in the cylinder is taken to be equal to the ambient temperature in which the filled cylinder must be kept for at least 8 hours. At a nominal pressure of 1.9 MPa (19.0 kgf / cm 2) at 20 ° C, the gas pressure in the cylinder in the temperature range from minus 5 to plus 40 ° C should be as specified in Table 6.
The residual gas pressure in the cylinder is measured with a pressure gauge of accuracy class 2.5 with a diameter of at least 100 mm according to GOST 8625. Cylinders from the consumer must come with a residual pressure corresponding to that specified in Table 7. Dissolved acetylene in cylinders is transported by all modes of transport in accordance with the rules for the transport of dangerous goods applicable to this type of transport, and the rules for the design and safe operation of pressure vessels. By rail, cylinders filled with dissolved acetylene are transported by car and small shipments in covered wagons. When transporting by small shipments, cylinder caps must be sealed. For the mechanization of loading and unloading operations and the consolidation of transportation by road transport medium-sized cylinders are placed in metal special containers. When transporting small-volume cylinders by all means of transport, they must be additionally packed in plank lattice boxes of type VII according to GOST 2991. Cylinders must be laid horizontally in boxes, with valves in one direction with mandatory gaskets between the cylinders, preventing them from hitting each other. Cylinders filled with acetylene are stored in special warehouses or in open areas under a shed protecting them from precipitation and direct sunlight, according to the coolant group 2 of GOST 15150. Safety requirementsAcetylene is an explosive gas. Acetylene explosions have great destructive power. With air it forms an explosive mixture with a lower concentration limit of ignition at atmospheric pressure, reduced to a temperature of 25 ° C, - 2.5% (by volume) according to GOST 12.1.004-85. Auto-ignition temperature 335 ° C. Auto-ignition pressure 0.14–0.16 MPa. Under certain conditions, acetylene reacts with copper to form explosive compounds; therefore, the use of alloys containing more than 70% copper is strictly prohibited in the manufacture of acetylene equipment. The pressure resulting from the explosion of acetylene depends on the initial parameters and the nature of the explosion. It can increase by about 10–12 times as compared with the initial one in an explosion in small vessels and increase when pure acetylene detonates 22 times, and when the acetylene-oxygen mixture detonates 50 times. Technical acetylene (with impurities) has a sharp unpleasant odor; prolonged inhalation causes nausea, dizziness and even poisoning. Acetylene has a narcotic effect. Poisoning is caused mainly by phosphorous hydrogen, which is in carbide acetylene. Gaseous acetylene is lighter than air and accumulates in the highest points of poorly ventilated rooms where the formation of an acetylene-air mixture is possible. The production of acetylene by fire hazard belongs to category A, according to classes of hazardous areas - to classes B1; B1a; B1b; V1g. The premises of acetylene production must have ventilation and exhaust ventilation. Compressed nitrogen, carbon dioxide fire extinguishers, asbestos cloth, sand should be used as fire extinguishing agents. Seamless acetylene cylinders are made of carbon and alloyed steel in accordance with GOST 949 - 73. Design features of cylinders (fig. 6.6). Acetylene cylinder has the same dimensions as the oxygen capacity of 40 dm 3. The mass of the cylinder without gas is 83 kg, the working pressure of acetylene is 1.9 MPa (19 kgf / cm 2), the maximum pressure is 3.0 MPa (30 kgf / cm 2). Fig. 6.6. Acetylene balloon: 1 - body; 2 - valve; 3 - nitrogen cushion; 4 - porous mass with acetone; 5 - shoe; 6 - safety cap Acetylene balloon is filled with a porous mass of activated charcoal, which is impregnated with acetone at the rate of 225 ... 300 g per 1 dm 3 capacity of the cylinder. Acetylene, dissolving well in acetone, becomes less explosive. Cylinders with a cast porous mass, capable of holding 7.4 kg of dissolved acetylene, are more economical, whereas cylinders with activated carbon are only 5 kg. On the cylinder with a molded porous mass below the inscription "ACETYLENE" the letters LM were painted with red paint. New cylinders are supplied with a nitrogen blanket. When acetylene is taken from the balloon, part of the acetone is removed as vapor. To reduce the loss of acetone during operation, it is necessary to arrange the cylinders in a vertical position and select acetylene at a rate not exceeding 1.7 m3 / h. In a filled cylinder with a capacity of 40 dm 3 at a working pressure and air temperature of 20 ° C, the volume of acetylene gas corresponding to normal conditions is 5.5 m 3. The color of the balloon is white, the inscription is red. Acetylene Cylinder Valve (fig. 6.7). The valve is made of steel. The use of copper alloys with its content of more than 70% is unacceptable, since explosive acetylene copper appears when in contact with acetylene.
Fig. 6.7. Acetylene cylinder valve: 1 - fitting for the socket wrench; 2 - the place of connection of the gearbox; 3 - shank with a tapered thread A distinctive feature of the acetylene cylinder valve is the absence of a flywheel and choke. In the valve body there is a side groove in which the acetylene reducer fitting is installed, pressing it with a special clamp through a leather gasket. This design of the valve does not allow the accidental installation of another gearbox in order to avoid the formation of an explosive mixture. Another distinctive feature of the acetylene cylinder valve is that it opens, closes and attaches the reducer to the cylinder with it using a special end wrench (Fig. 6.8).
Fig. 6.8. Special socket key acetylene balloon Determination of the volume of acetylene in the cylinder . The balloon is weighed before and after filling with gas, and the volume of gas in the cylinder is determined from the difference in performance and density of acetylene. Example. The mass of the cylinder with acetylene 89 kg, empty - 83 kg. The mass of acetylene in the balloon is as follows: 89 - 83 = 6 kg. The density of acetylene at atmospheric pressure and a temperature of 20 ° С is 1.09 kg / m3. Therefore, the volume of acetylene under these conditions is 6 / 1.09 = 5.5 m 3. Acetylene is widely used for gas welding and metal cutting. Not so long ago, it was obtained using a generator that ensures the breakdown of calcium carbide. But such an installation, despite all the measures taken, is characterized by increased danger. Therefore, acetylene is now increasingly being used in cylinders, which, among other things, is distinguished by its high purity, which allows welding and cutting to be performed more efficiently and efficiently. Acetylene propertiesAcetylene is a combustible gas, the mixture of which with oxygen allows the combustion temperature to be up to 3150 degrees Celsius. This substance is colorless and odorless (technical acetylene has a strong odor due to its impurities). Acetylene is practically insoluble in water, but in other liquids its solubility is quite high, especially in acetone (up to 28 liters of gas in 1 liter of liquid).
But the main danger associated with the storage of acetylene is its explosion hazard not only in mixture with air, but also in its pure form under certain conditions. At the same time, this gas releases much more thermal energy during an explosion than nitroglycerin or TNT (1.5 and 2 times, respectively). That is why it is impossible to store acetylene in standard conditions in a pure form. Acetylene cylinders
By volume, there are small (5 l), medium (10 l) and large (40 l) containers. The main difference is the internal filling of the cylinder. Since the cylinder with acetylene in a gaseous state is highly explosive, in practice the storage of gas dissolved in acetone is used. At the same time, in order to prevent the possibility of a backward impact of the flame and spontaneous decomposition of acetylene to an explosive state, a special filler is placed in the balloon. BAU-A (activated carbon) or LPM porous silica mass (cast porous mass) is used as a filler. This material takes up a third of the volume of the container, while the porous filler is able to absorb more gas. In order to ensure the explosion safety of acetylene is dissolved in acetone, which is filled with a cylinder with a porous filler. The amount of acetone is approximately 230 grams per 1 liter of the container capacity, and this is precisely what determines how much acetylene in the cylinder can be placed at full refueling. When the cylinder valve is opened, acetylene is evaporated, which is fed to the working devices. Acetylene cylinder requirementsCylinders for the storage of acetylene should be painted white, light gray paint is allowed, and they should have a red “ACETYLENE” inscription; in addition, if cast porous filler is used, the inscription “LM” is added.
The test is carried out at a pressure 1.5 times higher than the standard (35 MPa). In addition, every two years it is necessary to check the weight of the porous filler. The maximum allowable pressure of acetylene in the cylinder is regulated by GOST 5457-60 and depends on the ambient temperature. At 19 ° C, the pressure should not exceed 150 atmospheres (15 MPa), in most cases the cylinders are filled to 150 atm. It is forbidden to operate cylinders in the following cases:  Do not operate with strong heat. Violation of all these rules can lead to an explosion of acetylene. A few words about refilling cylindersThe amount of injected gas, and, consequently, the price of an acetylene balloon, is determined by simple weighing. The balloon is weighed before and after refueling, the difference in values is multiplied by 1.09 (weight of 1 cubic meter of acetylene at 20 degrees Celsius). The regulatory mass of an empty but ready for injection cylinder is knocked out in his passport. Approximately in a transport cylinder (40 liters) you can pump at least 5.5-7.5 kg of acetylene, into a 10-liter cylinder 1.4-2 kg, into a 5-liter 0.7-0.8 kg. In addition, more gas enters cylinders with a cast porous filler than in containers with activated carbon. In addition, it should be borne in mind that with each use of all the gas from a cylinder, about 150 grams of acetone, which must be replenished, comes out of it. Benefits of using acetylene in cylindersThe use of acetylene dissolved in acetone can significantly increase the performance of welding and cutting of metal. In addition, the use of acetylene cylinders has other advantages:
Despite the fact that the cost of acetylene in cylinders is somewhat higher, the economic effect of its use is significant, and it is explained by the ability to perform more work and the high performance of the equipment operating on this combustible gas. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Popular:
New
- How to rivet a rivet - automatic and manual methods for different materials What rivet rivet steel sheets
- Appointment of planar marking
- What should be used reinforcing mesh for concrete, wallpaper, plaster technology - plastic, fiberglass or metal
- Edit strip and sheet material
- Open lesson "editing, bending"
- Installation of staircases and platforms: general information
- Repair of pipes of water supply in the apartment Repair of steel pipes
- Markup definition. Planar marking. Types of markup. Questions for self-test
- Pipe bending machines Various variations of pipe bending machine
- Safety during filing




 The acetylene storage cylinder itself is almost the same as the oxygen one; it is also made from seamless steel pipe. A special design acetylene valve is installed on it, the fitting of which has no thread (the hoses are fixed with the help of a special clamp).
The acetylene storage cylinder itself is almost the same as the oxygen one; it is also made from seamless steel pipe. A special design acetylene valve is installed on it, the fitting of which has no thread (the hoses are fixed with the help of a special clamp).